by Image Permanence Instute
Rochester
written by James M. Reilly in 1993
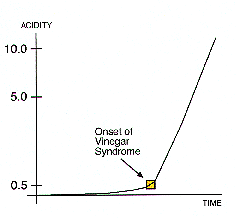
The reason why degradation follows this course is easy to understand. Acetic acid is formed when the acetyl “side groups” are split off from the cellulose molecules. There are three factors that induce such changes: heat, moisture and acid. In the early stages there is little acid present so the reaction rate is determined primarily by heat and moisture. During this long, slow buildup, the storage environment plays the decisive role. The temperature of the storage room determines how much heat energy is available to push deterioration along.
The sharp bend in the time-vs. acidity curve represents the point when acidity becomes another major factor, along with heat and moisture, in determining the rate of deterioration. The more advanced the deterioration becomes, the more the reaction rate is influenced by the presence of acidity. The reason is that the reaction is now “feeding on itself” – displaying what scientists call autocatalytic behavior. A catalyst is a substance that speeds up a chemical reaction but is not consumed by it. The whole process snowballs.
Image Permanence Institute
Rochester Institute of Technology
70 Lomb Memorial Drive
Rochester
NY 14623-5604
USA
