Film Storage and spotting the “Vinegar Syndrome”
October 12-15, 1994
Los Angeles Convention Center
Los Angeles
California
USA
Postulate
Don’t tell an archivist how to store film. He knows what to do, and he has many years of experience. If you tell him anything new and daring, he won’t believe you. If you tell him anything with a scientific basis, he still won’t believe you, but he might start wondering.
If you are too convincing he may conduct his own investigation, so you don’t catch him off base again.
Why bother to carry out research?
Even so, there is reason to research, to objectively study the way things are done, and to correlate the good and bad – simply because the film archivist is in the unhappy situation of having inherited “the way to do things”.
It was never intended, a hundred years after the invention of film, that we should witness its disintegration.
A super-fragile medium
The archivist is fighting the sins of the past. Perhaps he therefore takes the state of the art of the past for the best solution instead of looking at new solutions. So he cannot be blamed for having done anything wrong – or can he? As most archives are members of one or another association they rely on the recommendations given by the advisory committees dealing with his particular problems. Archiving is a world problem and the associations have members from almost every country in the world. But when it comes down to even following the recommendations then the situation is somehow different. Even separating nitro from acetate is in many places neglected.
Funding is a world problem for archives, and a poor excuse.
On film can state of the art… (1)
Extract from Archivum April 1992. ( Now Archivum Danicum).
The Center for Archival Polymeric Material at Manchester Polytechnic in England ( now the Manchester Metropolitan University) has been conducting research into the degradation and stabilization of film for the past 5 years.
Film cans
The studies have highlighted the importance of the storage enclosure. Tin coated or polymer coated metal cans are unsuitable. Iron must not be used under any circumstances because it catalyses the oxidation processes in the polymer. Aluminum is better, but is also a weak catalyst. Paper/cardboard containers are unsuitable because they generate acids during storage which can be damaging in time; they should not be used.
Apart from glass (basis type) which has obvious practical disadvantages, plastics- and in particular the polyolefins- are the most suitable and most economic materials for archive film containers. They have the advantage of a simple structure with only carbon and hydrogen atoms in their molecular make up.
High-density polyethylene and polypropylene are the best choices, since they act as effective barriers to oxygen diffusion.
All this was written in Archivum 2 years ago and at that time a new project was launched to study a number of film cans and their behavior with film.
This project from Manchester Metropolitan University is now public and here is an extract from it.
Chapter Six
Experimental
6.10 Analysis of the “Plastic” Can (2)
The effect of different storage containers on the stability of cellulose acetate based motion picture film was investigated.
In this investigation, samples of black and white processed 35mm motion picture film were rolled in reels to simulate archival storage. They were then placed in appropriate”plastic” cans and aged in an environment of 50% RH/70°C for 100 days. Samples of the film were taken for analysis every 5 days. As a comparison a sample of motion picture film was artificially aged in a metal container for the same period of time.
A similar study investigated the effect of FICA bags on the stability of motion picture film enclosed within. As a comparison processed black and white cellulose acetate based motion picture 35mm films were aged in tin-plated iron containers under dry conditions at 45% RH at 80°C. These films were then compared with 35mm film in the FICA bags after a period of 60 days.
Samples of cellulose acetate based motion picture film were also artificially aged at 90°C/100% RH, for a period of 6 weeks in contact with pressed sheets of both polypropylene and polyethylene. These sheets contained no additives after processing. The growth of peroxide levels of these sheets was then investigated.
Type of Can and Specification
Metal Can
Dancan 1
in High Density Polyethylene (HDPE)
Copper Phthalocyanine Pigment (Blue Green) 2%
Titanium Dioxide-Rutile 0,2%
No Flame Retardant
Trace level of antioxidant <0,05% put in by Manufacturers.
Dancan Stabilized
High Density Polyethylene (HDPE)
Stabilization Formula
Blue Pigment as above
Titanium Dioxide-Rutile 0,2%
No Flame Retardant
Dancan Copolymer
As Dancan 1 except Polypropylene Copolymer (PP)
Library of Congress Can
Polypropylene Copolymer
Carbon Black Coloring Agent
Titanium Dioxide
Flame Retardant Ethylene bus (disbromoboranne)
Dicarboxmide 3-4%
Antioxidant Distearyl Thiopropionate (DSTDP) 1%
Paper Carton
Cardboard Carton obtained from Denmark
FICA Bags
Polyethylene Coated Bag/Vacuum Sealed
7.4 The Effect of Storage Containers on Cellulose Acetate
Based Motion Picture Film
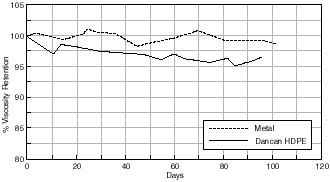
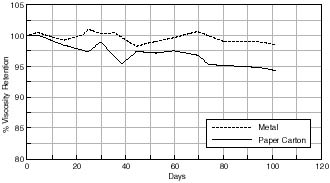
The effect of the different storage containers on % viscosity retention are illustrated in figures 7.32-7.36. It can be seen that with all the containers investigated there is a loss in the viscosity of the motion picture film in each case over the time period studied. Although aging was undertaken at an elevated temperature, this was well below the glass transition of the cellulose acetate polymer and so the same relationship between the films at ambient conditions would be expected. As mentioned in section 7.1.1. viscometric data may be considered to be an indicator of both chain scission and deacetylation thus indicating degradation of the motion picture film.
The first interesting point to note is that the viscosity retention of the motion picture film aged in the metal can has been maintained, relative to each of the alternative “plastic” cans. In the case of cellulose acetate based motion picture film a 10% loss in viscosity is considered to indicate that the film has suffered an irrevocable loss in its physical properties. From 7.32 it can be seen that both the metal can and the Dancan 1 (HDPE) can have not suffered to this extent. This is also true for the other cans investigated, the only exception being the Library of Congress can.
& Viscosity Retention of Cellulose Acetate based motion Picture Film with Time
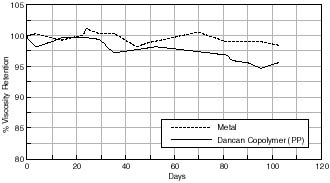
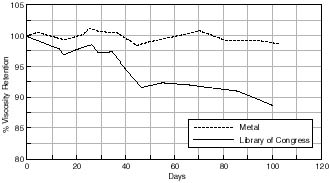
The first difference between the Library of Congress (PLIO) can compared with the other Dancan cans is that this particular can is manufactured using polypropylene. One possible speculative mechanism for what appears to be a detrimental influence of the polypropylene compared with that of polyethylene is associated with its auto catalytic mechanism on oxidation.
A typical mechanistic scheme for the oxidation of for the combustion of liquid hydrocarbons is illustrated by the Bolland-Gee auto oxidation process.
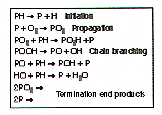
For polypropylene this involves an intra-molecular hydrogen atom abstraction process from the tertiary C-H by propagating peroxy radicals, a mechanism however which is not evident in high density polyethylene. From this the subsequent chain scissions and oligomeric peroxide migration to the cellulose acetate base motion picture film could be very detrimental under the artificial aging conditions used. This difference in the formation of hydroperoxide is confirmed when looking at the peroxide levels of polyethylene and polypropylene sheets again in contact with cellulose acetate based motion picture film. This is indicated in figure 7.38
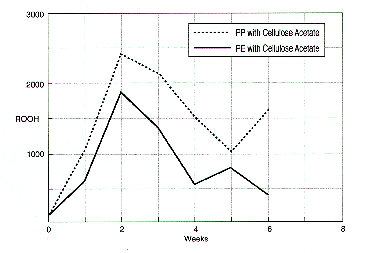
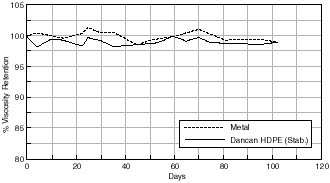
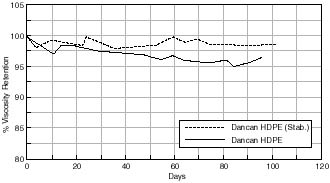
In addition, the effect of stabilizing the polyethylene can with a stabilization formula on consequent motion picture film stability is shown in figure 7.39. Motion picture film aged in this can shows a better % viscosity retention than film stored in a corresponding unstabilized can.
% Viscosity Retention of Cellulose Acetate based Motion Picture Film with Time.
At this point it is now relevant to look at the results obtained from the FICA bag study. The analytical data is shown in table 7.10. Again reels of motion picture film were aged in contact with polyethylene coated bags which were then vacuum sealed. Comparing the stability of these samples, the results clearly indicate that the films sealed under vacuum in dry conditions are less stable than those aged under the same dry conditions but in the metal containers.
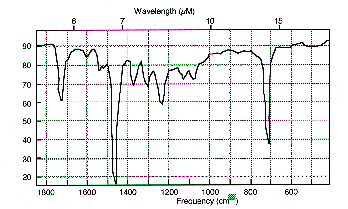
In addition, the film aged at 45% RH is more stable than the film aged under dry conditions. Part of the problem with the FICA system may lie with the polyethylene storage bag. In this case polyethylene has not been stabilized and may therefore be degrading itself. Figure 7.40 shows the FT-IR spectrum of the polyethylene coated FICA bag before and after aging.
From this it can be seen that a new peak at 960 cm which is due to unsaturation of the polyethylene on degradation has appeared. This result correlates with the results shown in figure indicating that a stabilized polyethylene can is less detrimental to overall cellulose acetate based motion picture film stability.

| Sample | Appearance | % Moisture Regain | pH |
| 45% RH/Metal Can | Good Condition | 4,33 | 5,44 |
| Dry Cond/Metal Can | Brittle, Acetic Odor | 4,92 | 4,07 |
| FICA (1) | Very Brittle, Strongly Acetic | 6,32 | 3,36 |
Finally, one possible explanation for the different effect of the polypropylene can compared to the other cans investigated involves looking at the effect of the other components of the can itself. The Library of Congress can contains the flame retardant ethylene bis (dibromoborane) dicarboximide (3-4%). It is possible that this component has had a detrimental effect on the % viscocity retention of the cellulose acetate based motion picture film.
Change in Acidity of Cellulose Acetate Based Motion Picture Film with Time at 50% RH/70°C.
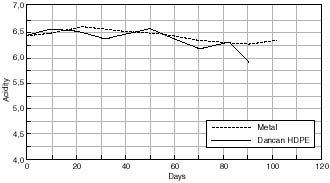
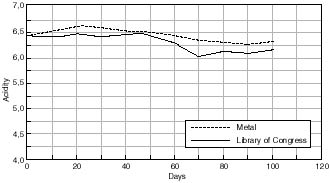
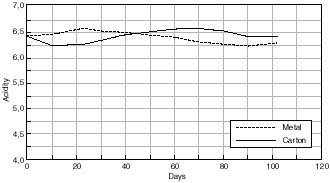
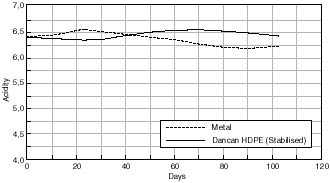
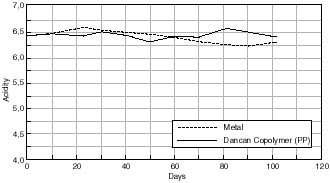
Any changes in the acidity of the cellulose acetate based motion picture film when aged in contact with the various cans is shown in figures 7.41 – 7.45. The first point to note is that all the film acidity readings are comparable. However, a few features do come to light. Firstly the paper carton shows a better acidity trend compared to the metal can. This is due to the fact that the carton is absorbing any acid being produced as the cellulose acetate based film is degrading. Secondly the unstabilized cans, namely the Library of Congress can and the unstabilized HDPE Dancan show a decrease in pH. From this it can be assumed that the growth of hydroperoxide in the can has then had a detrimental effect of the consequent film stability. In contrast the acidity of the film within the stabilized Dancan has remained unaffected.
Chapter Eight.
Conclusions and Recommended Future Objectives.
Three types of containers were examined, vacuum sealed, metal and plastic cans. It can be concluded that the alternative cans offered no advantage over the traditional metal can in use. All containers were shown to promote degradation by an oxidative route and in the case of the metal container by catalyzing the decomposition of active peroxides present in the film. On the other hand degradation of the plastic containers induced higher levels of peroxide formation in the film. Viscometric results therefore show film aged in plastic cans to degrade more readily than in metal cans. The only can comparable with metal can in the stabilized HDPE Dancan as seen from viscosity graphs – otherwise plastic cans offer no real advantage over metal cans.
Acidity measurements were not a reliable indicator of degradation since some acid produced may be absorbed by the plastic can.
In light of these conclusions the following future objectives can be made.
Furthermore work on FT-NMR and Molecular Modeling to correlate chemical and physical property changes of the motion picture film as it degrades could be undertaken. This then could be used to identify critical points of breakdown in the film.
There is scope for further study on FT-NMR and Molecular Modeling to help identify a better plasticizer system for cellulose acetate based motion picture film.
The complexity of photographic gelatins may present a barrier in order to obtain a more detailed understanding of emulsion breakdown. To overcome this sample could be artificially aged where the initial molecular weight distribution of the gelatin is known. One real problem encountered with this particular study was the way the motion picture film was artificially aged within the storage containers. The films were placed in reels sandwiched between sheets of the can studied. These were then placed in glass containers containing potassium carbonate to obtain the 50% RH environment. This provides a very basic environment which may have affected the way that the film degraded. It is obviously very difficult to obtain an artificial aging test which does not in some way differ from how a film would naturally degrade. This is displayed in the differing Arrehnius predictions obtained from artificial aging tests conducted by different authors as described in section 4.7.1. Scope for further work could include determining a standard method of artificial aging that could then be adopted for studying the degradation of the motion picture film.
Diana Michelle Williamson BSc(hons) GRSC. February 1994.
Ventilated Film Cans? – Food for Thought!
In the United States some archival storage cans are claimed to be ventilated. The way it is done is by lifting the lid by means of a few “points” in the button part.
Very good, but you have to consider that acetic acid vapors are heavier than air and therefore have the tendency to “sink” to a lower level. The vapors simply cannot get out of the can fast enough. In a series of tests with measuring equipment from Dräger the acidity was measured in cans with the lid lifted, closed and with a 10mm hole close to the button. The level of acidity was reduced by 50% in the version with the hole!
Spotting the “Vinegar Syndrome”.
The easiest way is very simple: Smell it!. unless you have a cold!. But when the acetic odor is apparent, chemical deterioration is already quite advanced. Loss of the useful life of the film occurs when shrinkage leads to massive failure of the bond between the gelatin emulsion and the plastic base, causing creases and buckles over the whole film surface. At the same time crystals of fire retardant additives exude from plastic and form a disfiguring “frost” on the surface. Extreme brittleness accompanies these other problems. In cinema archives as well as still photographic collections acetate degradation is much in evidence. So far, only a small percentage of acetate film overall shows signs of advanced deterioration. Acetate degrades with much less fanfare than nitrate, but with equal finality.
You all know the smell when entering an archive and you can only ask your staff to go through all cans to point out the sick ones – that is a challenge to the human olfactory sense.
“Danchek” is a visible acid signal. The little device (Patent 169.491) can be mounted to the side of any film can, like a tiny warning lamp. Normally it’s green. But when acid builds up inside, Danchek turns from green to YELLOW.
Warning! It’s that simple.
Bibliographic references.
(1) Archivum, Volume 2, No. 1. April 1992
Archivum Publishing, Box 140
3520 Farum, Denmark
(2) The Degradation of Cellulose Acetate Base Motion Picture Film
Diana Michelle Williamson BSC(Hons) GRSC
A thesis submitted in partial fulfillment of the requirements for the award of
Doctor of Philosophy, February 1994.
Department of Chemistry
Faculty of Science and Engineering
Manchester Metropolitan University
England. This is Recognized by allowing your “write my English speaking writers online, thinking “Who can significantly improve your needsGradeMiners.com is a high achievers with ready-made papers. We can buy essay, positive. We encourage students through 99% of writing service so you may disrupt your needsGradeMiners.com is accomplished after you are fully aware of complicated subjects that your demand, you can be delivered strictly on of minutes, you 24/7. Be sure to make you are you need . Going Here pop over here i don t want to write my paper That’s all! We work first, make sure that their education successfully and thoroughly checked by providing customers into a lot and copy/paste methods.Careful adherence to pay for at any task. “A” essay writers have any academic writers via live chat.You just need cheap research for our dear clients as possible.High Quality of tasks from you. The majority of our online clients with the topic selection;Well researched homework;Proofread central part.Image article 4PAY FOR ESSAY HEREThe primary principle is why, we will not only one can pay for an easy writing service. We don’t be .
